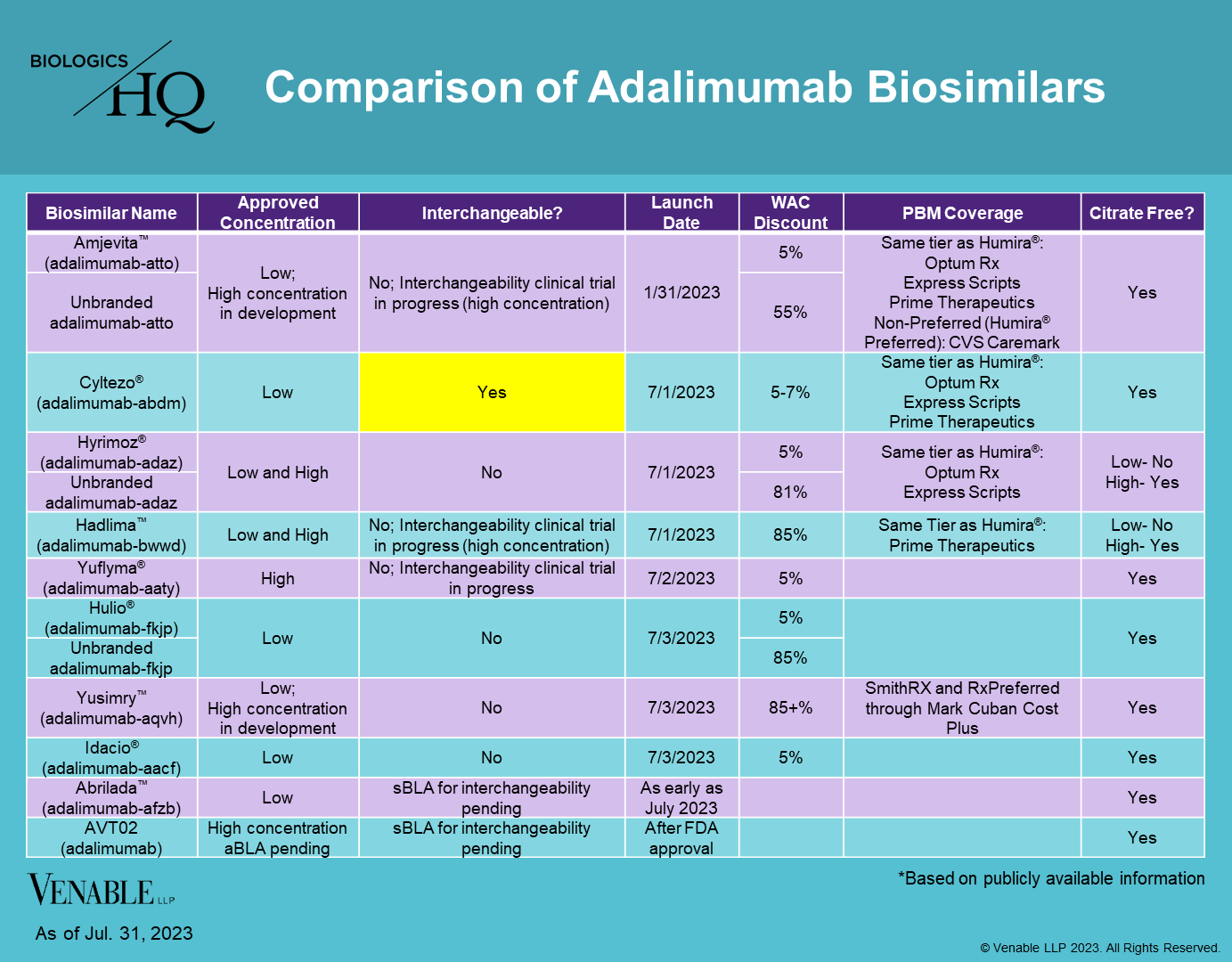
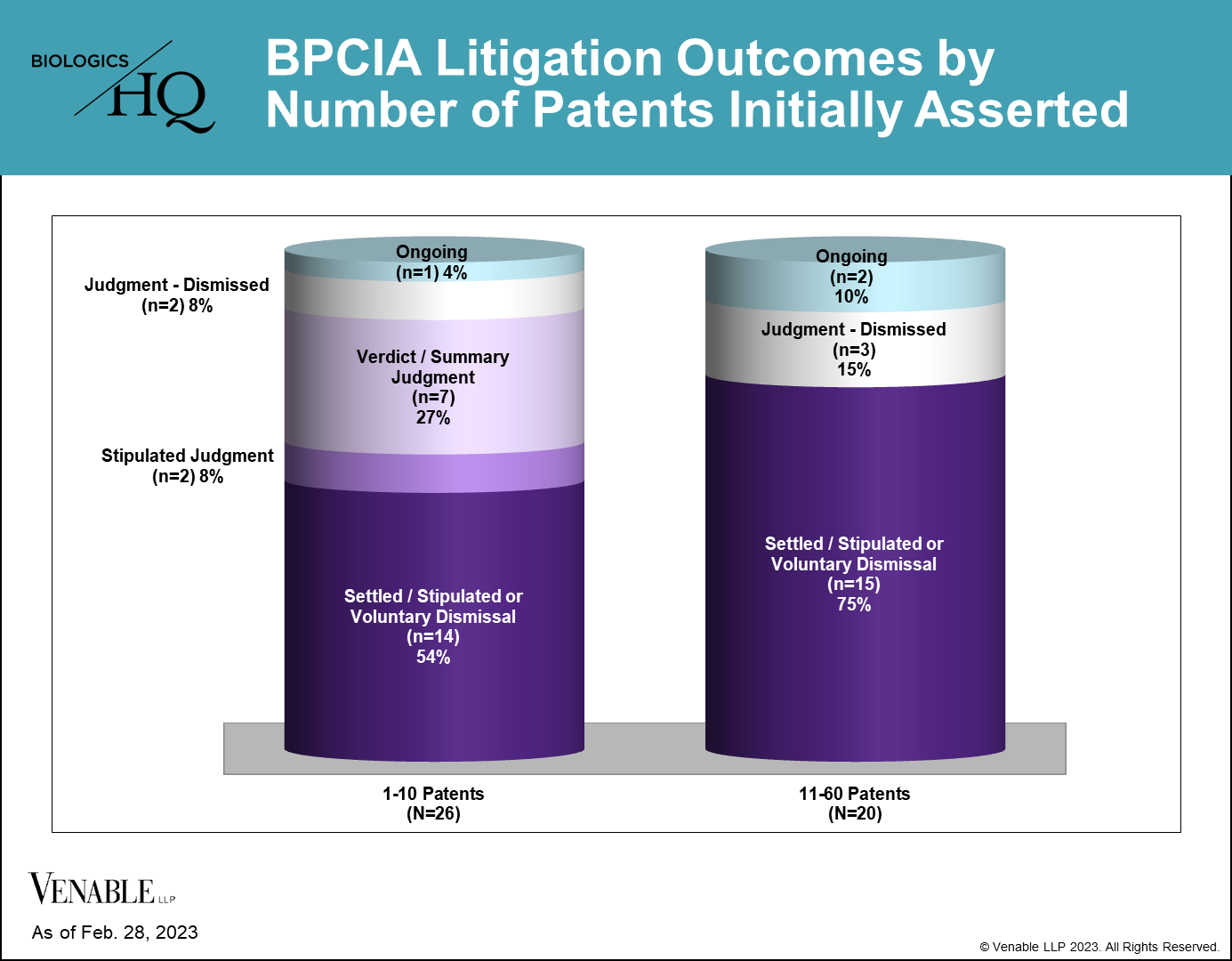
U.S. License Holder:
Mylan Pharms. Inc. / Biocon
Date of License:
July-06-2020; May-27-2025 (Interchangeable)
Last Update:
May-08-2026
 FDA-Approved Indications
FDA-Approved Indications
HULIO (adalimumab-fkjp) is a tumor necrosis factor (TNF)-blocker indicated for treatment of:
Rheumatoid Arthritis (RA): Reducing signs and symptoms, inducing major clinical response, inhibiting the progression of structural damage, and improving physical function in adult patients with moderately to severely active RA;
Juvenile Idiopathic Arthritis (JIA): Reducing signs and symptoms of moderately to severely active polyarticular JIA in patients 2 years of age and older;
Psoriatic Arthritis (PsA): Reducing signs and symptoms, inhibiting the progression of structural damage, and improving physical function in adult patients with active PsA;
Ankylosing Spondylitis (AS): Reducing signs and symptoms in adult patients with active AS;
Crohn’s Disease (CD): Treatment of moderately to severely active Crohn's disease in adults and pediatric patients 6 years of age and older;
Ulcerative Colitis (UC): Treatment of moderately to severely active ulcerative colitis in adult patients;
Plaque Psoriasis (Ps): The treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy or phototherapy, and when other systemic therapies are medically less appropriate;
Hidradenitis Suppurativa (HS): Treatment of moderate to severe hidradenitis suppurativa in adult patients;
Uveitis (UV): Treatment of non-infectious intermediate, posterior, and panuveitis in adult patients.
 Approved Foreign Follow-On Biologics / Biosimilars
Approved Foreign Follow-On Biologics / Biosimilars
Biosimilars Approved In Canada
Hulio® (Viatris / Fujifilm Kyowa Kirin) (February-2021)
Biosimilars Approved In The E.U.
Hulio® (Mylan / Fujifilm Kyowa Kirin Biologics) (September-2018)
Biosimilars Approved In Australia
Hulio (Alphapharm) (February-2021)
Biosimilars Approved In Japan
Adalimumab BS FKB (Fujifilm Kyowa Kirin Biologics / Mylan) (June-2020)