Spotlight On: Humira® (adalimumab) / Amjevita™ (adalimumab-atto) / Cyltezo® (adalimumab-adbm) / Hyrimoz™ (adalimumab-adaz) / Hadlima™ (adalimumab-bwwd) / Abrilada™ (adalimumab-afzb) / Hulio® (adalimumab-fkjp) / Yusimry™ (adalimumab-aqvh) / Idacio® (adalimumab-aacf) / Yuflyma® (adalimumab-aaty) / Simlandi® (adalimumab-ryvk)
by April Breyer Menon; Corinne E. Atton ; Joseph K. Yarsky ; Robert S. Schwartz, Ph.D.
April 30, 2026
Download PDF
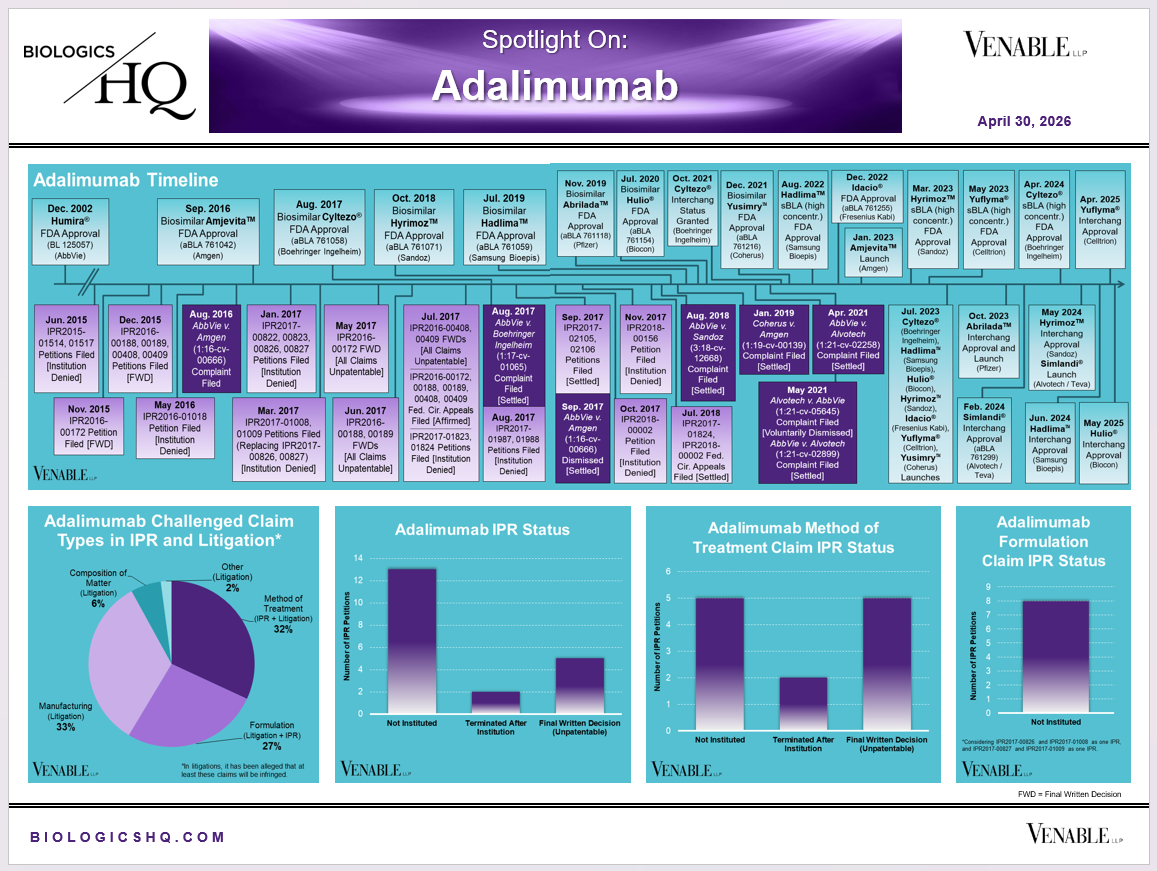
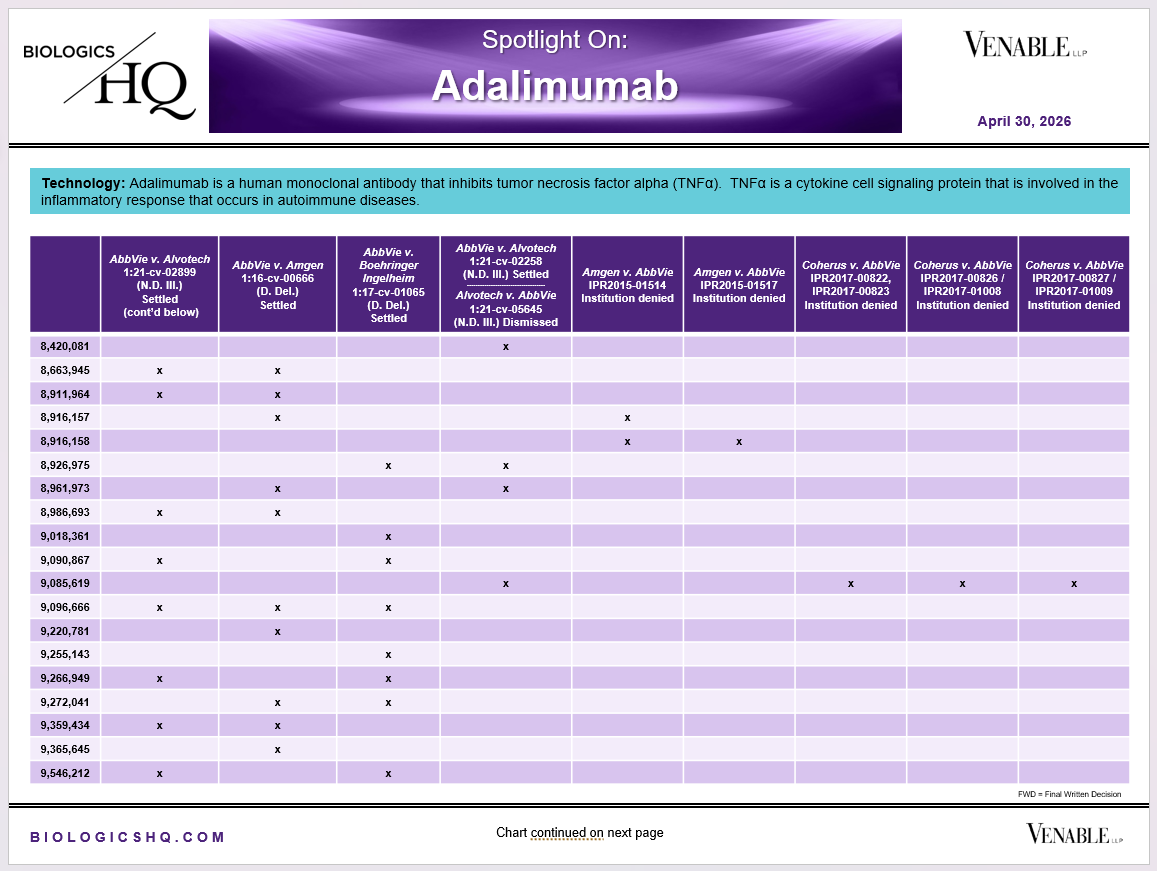
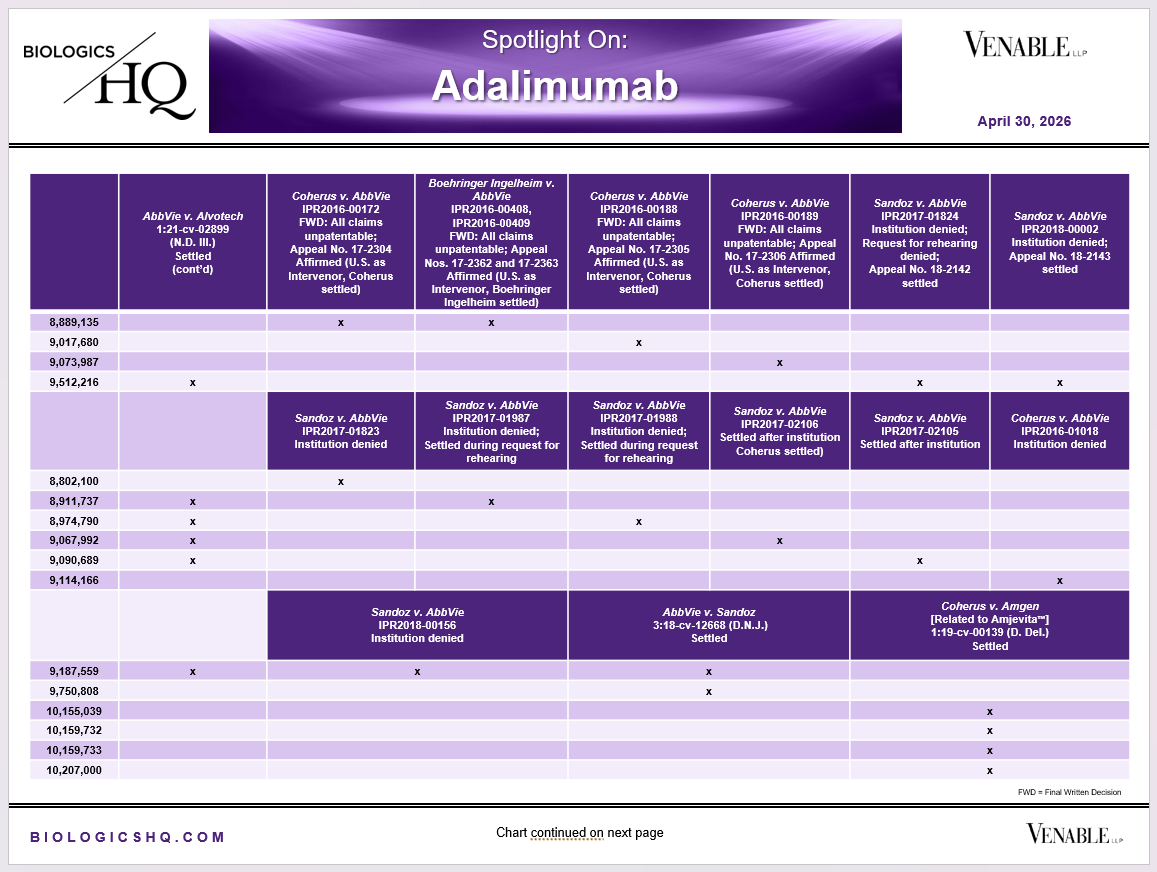
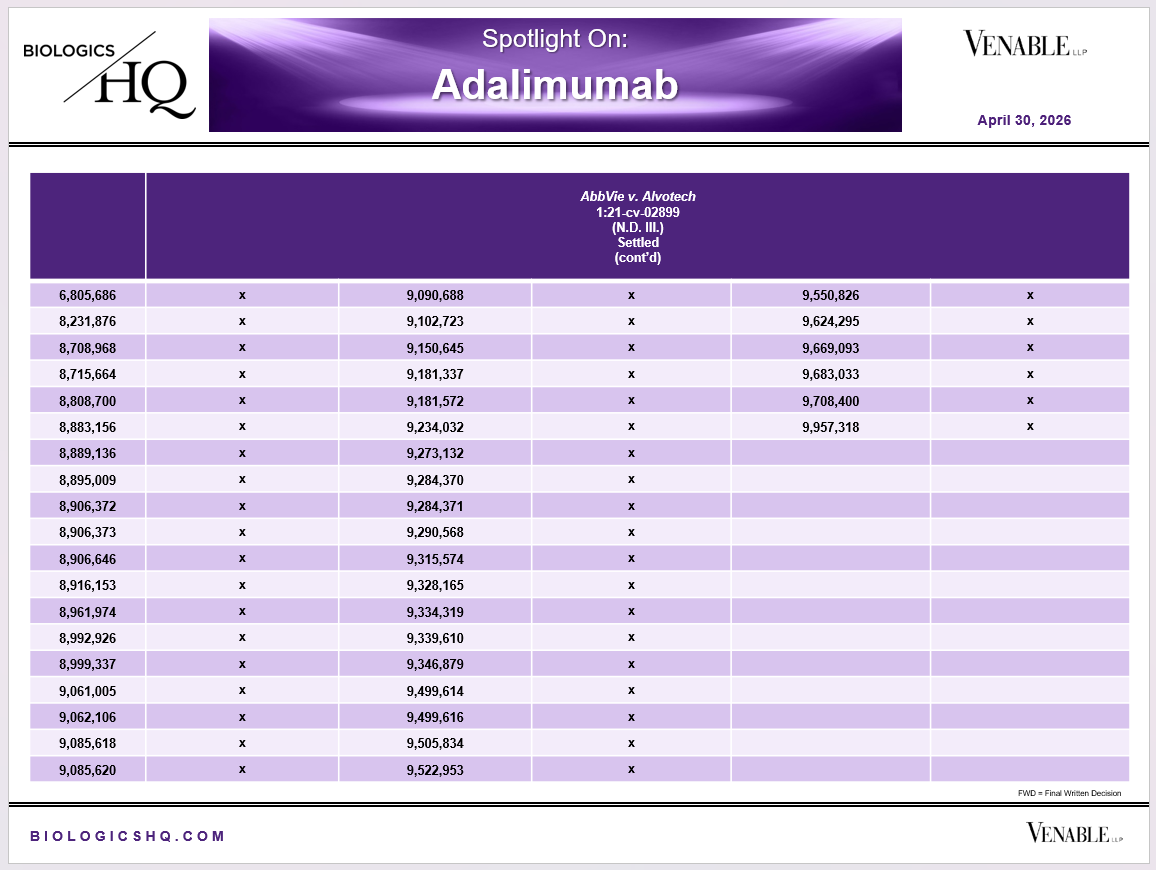
*Adalimumab Challenged Claim Types in IPR and Litigation: Claims include those challenged in litigations and IPRs. Claims are counted in each litigation and IPR, so claims from the same patent challenged in multiple litigations/IPRs are counted more than once. Within each litigation a claim is counted only once. Within each IPR, claims are counted only once, whether they are challenged under § 102, § 103, or both. IPR2017-00826 and -01008 are counted as one IPR, IPR2017-00827 and -01009 are counted as one IPR. Claims in litigations are determined based on claims alleged infringed in Complaints.
Click on the “Subscribe” link above for access to future product dashboards and updates.






