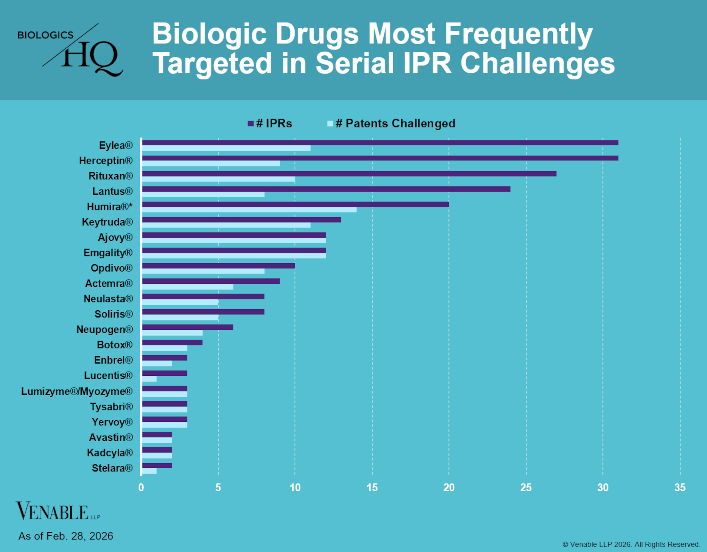
by April Breyer Menon | Mar 4, 2026
Biologic Drug IPR Petitions include IPR petitions relating to CDER-listed / 505(b)(2) biologic products. IPR petitions relating to manufacturing patents that may be relevant to multiple products (for example, U.S. Patent No. 6,331,415 (a “Cabilly” patent)) are...
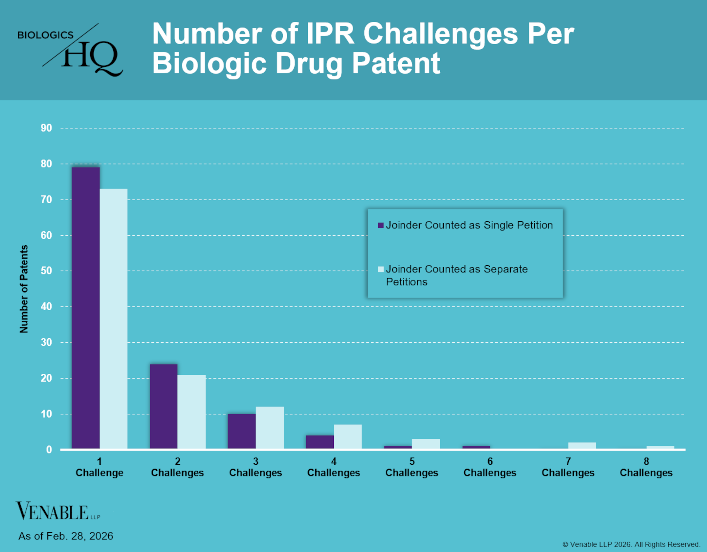
by April Breyer Menon | Mar 4, 2026
Biologic Drug IPR Petitions include IPR petitions relating to CDER-listed / 505(b)(2) biologic products. IPR petitions relating to manufacturing patents that may be relevant to multiple products (for example, U.S. Patent No. 6,331,415 (a “Cabilly” patent)) are...
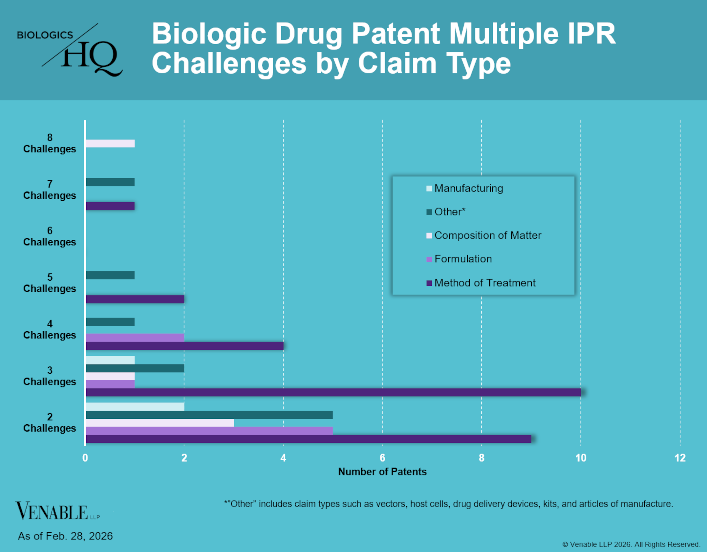
by April Breyer Menon | Mar 4, 2026
Biologic Drug IPR Petitions include IPR petitions relating to CDER-listed / 505(b)(2) biologic products. IPR petitions relating to manufacturing patents that may be relevant to multiple products (for example, U.S. Patent No. 6,331,415 (a “Cabilly” patent)) are...
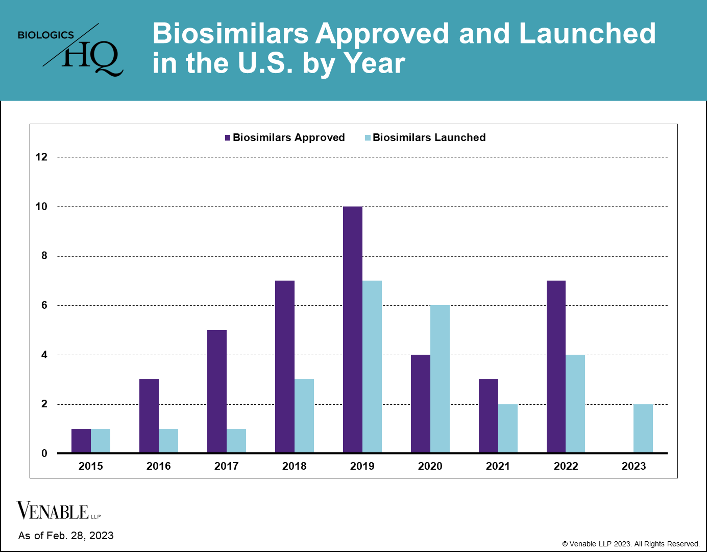
by April Breyer Menon | Mar 17, 2023
As of February 28, 2023, there were 40 approved biosimilars of 11 reference products in the U.S. market, seven of which were approved in 2022. This is a marked increase over the prior two years, which had seven total approvals due to COVID-19 related approval...
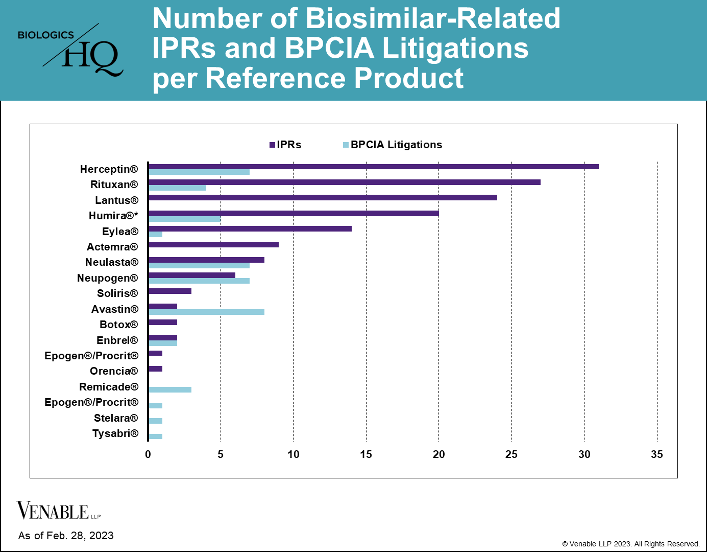
by April Breyer Menon | Mar 17, 2023
*One litigation is counted twice as it related to patents covering both Neupogen and Neulasta. While Lantus patents have been subject to litigation, none of those litigations were under the BPCIA and thus are not included. As of February 28, 2023, there have...







